A few months ago, a paper was published called Macroscopic heat release in a molecular solar thermal energy storage system available here: https://pubs.rsc.org/en/content/articlehtml/2018/ee/c8ee01011k
The overall concept is explained in this one sentence: “A solar thermal fuel is like a rechargeable battery, but instead of electricity, you put sunlight in and get heat out, triggered on demand,” – Jeffrey Grossman, Engineer MIT
Here is a simple diagram that shows the concept:
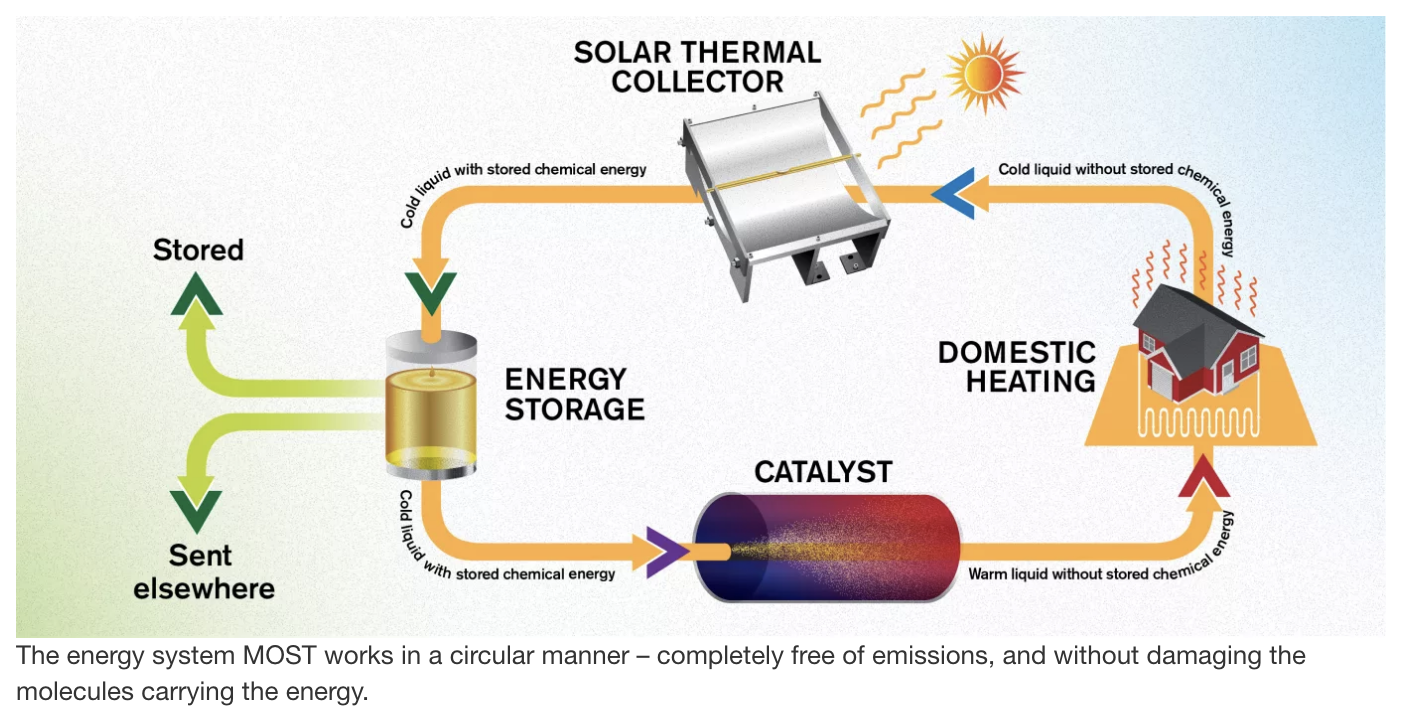
When heat from the sun warms a fluid, it changes the molecules so that this heat can be released at a later time on demand when it is exposed to a catalyst.
What I see as a huge instant benefit is the efficiency of the system. Solar panels are around 20% efficient, which is horrible, but some of the best solar heat collectors such as the evacuated tube collectors can be in the high 90%. Being that heating accounts for about half of the average home energy use, this method makes a lot of sense.
It is likely you have experience with a fluid that operates similar to this – sodium acetate heating pads. These are plastic containers with a simple chemical solution. When you heat them up, the solution liquefies and stays that way until activated. There is a tiny metal, flexible disc inside. When the disc is bent or “clicked”, it triggers a chain reaction crystallization process of the chemical that heats up. This is of course oversimplified compared to the Solar Thermal Fuel technology, but is suffice to convey the concept in simple terms.
For an article, which is much simpler than the paper listed at the top of this post, go here: https://www.sciencealert.com/scientists-develop-liquid-that-sucks-up-sun-s-energy


