A university project created a cooling system that creates about 600% more work than it takes to run the system using memory metal.
A university project created a cooling system that creates about 600% more work than it takes to run the system using memory metal.
In this brief article, you’ll learn how something can easily produce way more work than it takes to run it and there are no violations of physics.
Electric heating element heaters are energy hogs for the heat they produce but regardless, they are 100% efficient already. And there is nothing magical or special about a 100% efficient heating system.
Why? Because nichrome or other heating element wire is thought to dissipate or waste all the electrical current that runs through it. That is not even close to being the reality, but that is what conventional science believes so we’ll just entertain that for the purposes of this article and to keep it simple. Normally, heat is considered waste in non-heat systems but if you power a heating element and it “wastes” all the electricity, then it is 100% efficient since the desired work is the heat.
Efficiency is a funny subject because as absolute as it sounds, it is really an objective measurement because efficiency changes depending on your perspective of what kind of work you want.
An incandescent bulb is considered 10% efficient since the desired work is light while 90% of the energy is “wasted” in heat. However, if we want to use those bulbs as heaters, then magically, they would be considered 90% efficient, with 10% wasted as light. But if you want some heat AND light, then the bulb is also magically considered 100% efficient since 100% of what you are using to power the bulb is creating both power and light.
Heat pumps create hundreds of percent more heat/cooling than the electricity required to run them – geothermal, refrigerators, air conditioners, air source heat pump dryers and water heaters, etc. yet, they are less efficient than heating element heaters.
Efficiency is a ratio of TOTAL input to TOTAL output. The heating element produces as much heat as the wattage is provided. It is 100% efficient.
But there is another measurement, COP or Coefficient of Performance, which is the ratio of ONLY WHAT WE PROVIDE on the input compared to the TOTAL output. With a heating element, we provide all the input so total output divided by our input will equal 1.0 or COP of 1.0.
With heat pumps, the story is very different. If it takes 200 watts of electricity to run a compressor to circulate a refrigerant through compressions and expansions cycles (that creates high and low potentials in the system), heat moves for free towards a colder area so the total amount of heat moved in a heat pump can easily be 300, 400, 500, or 600 watts worth of work in an electrical equivalent – for only paying 200 at the wall.
If you put in 200 but you get 600 watts of heat movement, the total desired work is 600 watts worth divided by only your input (not counting free environmental contribution) of 200 watts = 3.0 or COP of 3.0. That is a 300% NET GAIN in total energy (work) produced compared to what you have to pay for. But if you look at all input including environmental input, it will be under 100% since there are losses. That is how you can have a system that produced hundreds of percent more work compared to what you pay for although it will still be 100% efficient of less.
The cooling system methodology created by a team led by Professors Stefan Seelecke and Andreas Schütze at Saarland University is technically not new in concept, but it is the best example of taking advantage of an interesting memory metal called Nitinol and it’s known effects of soaking up heat while it gets bent and releasing the heat when it straightens out.
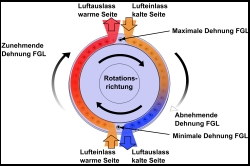
The cooling system is made of a cylindrical chamber with a rotor that has nitinol metal strings running along the length. There is a cam system that flexes the wire as it rotates for 1/2 the revolution. During this 1/2, the wires soak up heat in that compartment, which cools the compartment down. When the wires goes into the other side of the chamber, they are allowed to straighten up and they release a lot of heat, which heats up that compartment.
There is air moving through the devices to move both the hot and cold air. The claims from the university is that it is about twice as efficient as a heat pump. As you know from the examples above that a heat pump could have a COP of 2.0 or 3.0 easily. That means that this new Nitinol cooling device would have a COP of 4.0-6.0 or 400-600% more work done than the motor takes to rotate the cylinder!
Although the recent buzz has been talking about this latest development as if it is new, but the university has been at this project for several years. Read this to see where their thought process came from as well as their funding. https://www.asminternational.org/web/smst/newswire/-/journal_content/56/10180/26145309/
It also has this graphic that illustrates a bit of how it works – this is not shown in any of the recent media releases.
Here is a PDF of the German patent: http://emediapress.com/wp-content/uploads/2019/04/DE102016118776A12.pdf
Here is the PDF with a low quality translation to English – good enough to understand what is going on: http://emediapress.com/wp-content/uploads/2019/04/DE102016118776A11.de_.en_.pdf
Who will be the first to replicate the claims and post their results in http://energeticforum.com?
For more info on Nitinol, go here: Nitinol
To learn how open systems are permissible by the laws of physics and that systems CAN and DO produce more work than it requires from the operator by getting these two presentations:
Hacking the Aether by Aaron Murakami: https://emediapress.com/shop/hacking-the-aether/
Open System Thermodynamics by Peter Lindemann: https://emediapress.com/shop/open-system-thermodynamics/
Share this article with your friends using the links below – let’s get some replications going!
Update: March 31, 2025 – The Nitinol Cooling Revolution Advances
Since I wrote about the Nitinol-based cooling machine from Saarland University six years ago, claiming a remarkable 400-600% overunity—or Coefficient of Performance (COP) of 4.0–6.0—based on its edge over traditional heat pumps, the story has taken some impressive turns. In 2019, I spotlighted this German innovation, highlighting how Nitinol’s shape-memory properties could redefine refrigeration. Now, with fresh developments from Saarland and beyond, it’s time for an update. This covers the latest progress, clarifies the science, and tracks a global push in elastocaloric tech—complete with the critical distinction between COP and efficiency that too many still miss. Here’s the rundown.
The Starting Point: 2016–2019 Recap
In 2016, Saarland University’s Stefan Seelecke and Andreas Schütze introduced a prototype that caught my eye: a rotating cylinder with Nitinol wires, flexing to absorb heat and releasing it elsewhere, driven by a small motor. The ASM International article (Shape memory Nitinol alloys designed to cool refrigerators) set the stage, but their 2019 piece (Saarland University refrigerator is based on heating and cooling Nitinol wire bundles) delivered the key claim: it operated “at twice the efficiency of a heat pump.” With heat pumps at COPs of 2.0–3.0, I estimated 4.0–6.0. This wasn’t perpetual motion—thermodynamics holds—but a standout heat-moving system, backed by a German patent (DE102016118776A1, English PDF). Material COPs were already hitting 7.0–14.9 in studies like APL Materials (doi:10.1063/1.4952980), signaling big promise.
2022–2025: Saarland’s Steady Progress
By 2022, Saarland showcased a continuous-operation prototype at Hannover Messe, using fine Nitinol wires (200 microns) that stretch to cool (up to 20°C drops) and relax to heat, with airflow managing the temperature shift. The NEKKA project, launched in January 2022 with €6 million from Germany’s Federal Ministry of Economics, targeted electric vehicle (EV) cooling, allocating €1 million to Seelecke’s team. At Hannover Messe 2023, they pitched it as “the most promising alternative” to conventional refrigeration, a view echoed by the EU and U.S. Department of Energy.
The breakthrough came in April 2024 with a mini-refrigerator prototype (ScienceDaily), unveiled at Hannover Messe. This compact unit, cooling a single bottle, flexed Nitinol bundles for a 20°C span, boasting a COP “more than ten times” that of typical systems. If conventional ACs or heat pumps sit at 2.0–3.0, that’s 20.0–30.0+—a massive jump from my 2019 estimate. No exact figures yet, but X posts from Hannover 2025 (March 31) hint at COPs possibly reaching 15–25. This reflects refinements: thinner wires, optimized cycles, and improved heat transfer. System COPs in papers like Nature Communications 2023 (doi:10.1038/s41467-023-43611-6) hit 3.7, but this suggests they’re nearing material peaks (7.0–20.0).
Beyond Saarland: Global Momentum
The elastocaloric field is gaining traction worldwide. A 2023 Science paper (doi:10.1126/science.adg7043) from the University of Maryland achieved a 50.6K temperature lift with Nitinol, though system COPs ranged from 1.03–6.85. In August 2024, Hong Kong University of Science and Technology (HKUST) upped the ante with a multi-material Nitinol setup, hitting a 75K lift—that’s a 75 Kelvin temperature swing, or about 135°F—and 48% better efficiency than prior benchmarks, pushing material COPs likely past 20. These advances pressure Saarland to innovate, with NEKKA eyeing EV applications into 2025.
COP vs. Efficiency: Setting It Straight
Let’s clear this up: COP and efficiency aren’t the same, despite widespread confusion. Efficiency is work output over total energy input, maxing at 100%. COP is heat moved per work input, and it can soar past 1.0 by tapping ambient energy. A heat pump with a COP of 3.0 shifts three units of heat per unit of power—not 300% efficient, just highly effective. My 2019 “400-600% overunity” meant COP, not a physics-defying claim. That 2024 “ten times more efficient” line? It’s COP 20.0+, not efficiency, despite the vague phrasing. Material COPs of 14.9–20, as seen in a 2021 International Journal of Refrigeration study (doi:10.1016/j.ijrefrig.2021.03.017), support this, but system COPs hitting that mark is the game-changer—stunning if verified.
Milestones and Hurdles
From 2016’s concept to 2024’s mini-fridge, Saarland’s notched achievements: COPs doubling heat pumps (4.0–6.0) by 2019, then potentially tenfold gains (20.0+) by 2024. Material COPs over 7.0 were documented by 2016 (PMC PMC4942021), with system COPs climbing—3.7 in 2023, maybe 20.0+ now. Hurdles remain: Nitinol’s cost, cycle fatigue, and scaling to practical sizes. Yet, Germany’s 2023 Energy Efficiency Act could drive adoption in data centers or EVs.
Looking Ahead
The trajectory’s clear—2019’s “twice as good” evolved to 2024’s “ten times better,” with material COPs leading and system designs catching up. Saarland’s NEKKA and mini-fridge milestones, alongside global efforts, point to a refrigeration shake-up. Will COP 20.0+ cool your home soon? Not quite, but Hannover Messe 2025 (unfolding now!) might reveal more. This technology’s on the cusp of something big—stay tuned!
References/Resources
- Original Article: 400-600% Overunity Cooling Machine
- 2016 ASM Intro: Shape memory Nitinol alloys designed to cool refrigerators
- 2019 ASM Claim: Saarland University refrigerator is based on heating and cooling Nitinol wire bundles
- Patent: DE102016118776A1 English
- Material COP 14.9: APL Materials 2016, doi:10.1063/1.4952980
- System COP 3.7: Nature Communications 2023, doi:10.1038/s41467-023-43611-6
- Material COP 7.0: PMC 2016, PMC4942021
- 2024 Mini-Fridge: ScienceDaily
- NEKKA Project: ScienceBlog
- Global Lift 50.6K: Science 2023, doi:10.1126/science.adg7043
- HKUST 75K: EurekAlert (Aug 2024)
- Material COP 20: International Journal of Refrigeration 2021, doi:10.1016/j.ijrefrig.2021.03.017 (check journal access)
- Early Microcooling: Nature Energy (2016)
Bibliographical Notes
- Ossmer, H., & Kohl, M. (2016). Elastocaloric cooling: Stretch to actively cool. Nature Energy.
- Qian, S., et al. (2023). High-performance elastocaloric cooling. Science.
- Seelecke, S., et al. (2016). Elastocaloric cooling processes. APL Materials.
- Tušek, J., et al. (2021). Elastocaloric effect in NiTi alloys. International Journal of Refrigeration.